Dimensions: The most comprehensive view of the research landscape
Accelerating insight from scientific literature
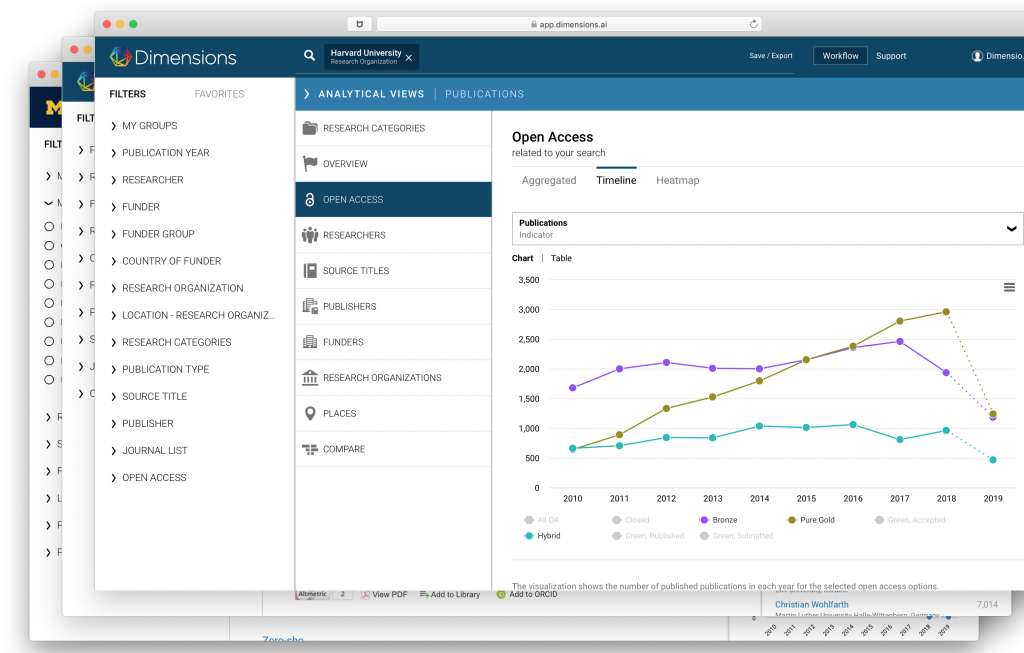
Rapid discovery and evaluation
Having access to the world’s largest connected research database is just the beginning…
Dimensions layers our comprehensive collection of data with easy-to-understand visualizations and integrated AI technology that speeds up analysis and decision making.
The result — a powerful search tool with discovery capabilities that speeds up time to insight .

An innovative solution for every research need
Our combination of data breadth and depth with cutting-edge technology was built to speed up time to insight.
What makes Dimensions special?
- Access to the world’s largest connected research database, covering publications, conference abstracts, grants, patents, clinical trials, datasets, policy documents and more.
- Full-text search across our database, beyond title and abstract, ensuring that you surface more relevant information than anywhere else.
- Built in AI technology to speed up time to insight – whether that’s for finding what you need or understanding your results.
- Purpose-built visual applications that help you quickly perform specific tasks, tailored to the needs of academic institutions and their researchers, government organizations and agencies, nonprofits, publishers, and life sciences and R&D departments – whatever your focus might be
Dimensions enables you to search, extract and analyze the research data you need, whenever you need it, how you need it.
Search features
Full text search for more than 200 million documents, with metadata filters, similarity search, and advanced features that enable you to structure the search in the way you want, and define your own groups of entities.
Data analysis
Aggregate and visualize data within the application itself, with heatmaps, VOSviewer and more. Use impact metrics based on Citations data / Altmetric Attention Score are also available.
Data export
Export Dimensions metadata – for analysis, to build VOSviewer networks, to import into reference managers, or to create result sets for Dimensions’ dashboard apps.
Researcher workflows
Manage ORCiD records, ReadCube Papers integration, BibTeX/Ris exports, and more.
Full-text linking
Faster access to published research with Open Access linkouts based on Unpaywall, integration with GetFTR & LibKey, and OpenURL resolver support.
Integration with other Modules&Apps
Analytical and workflow apps for different users and use cases – all drawing on the world’s largest linked research information dataset.
Custom implementations
Proprietary data such as grant applications can be enriched and securely integrated into a custom environment, for further analysis and reviewer identifications.
Support & training
Our 24/7 support team is always available to help.
Explore Dimensions
Dimensions has different options, so you can choose the version that’s best for you.
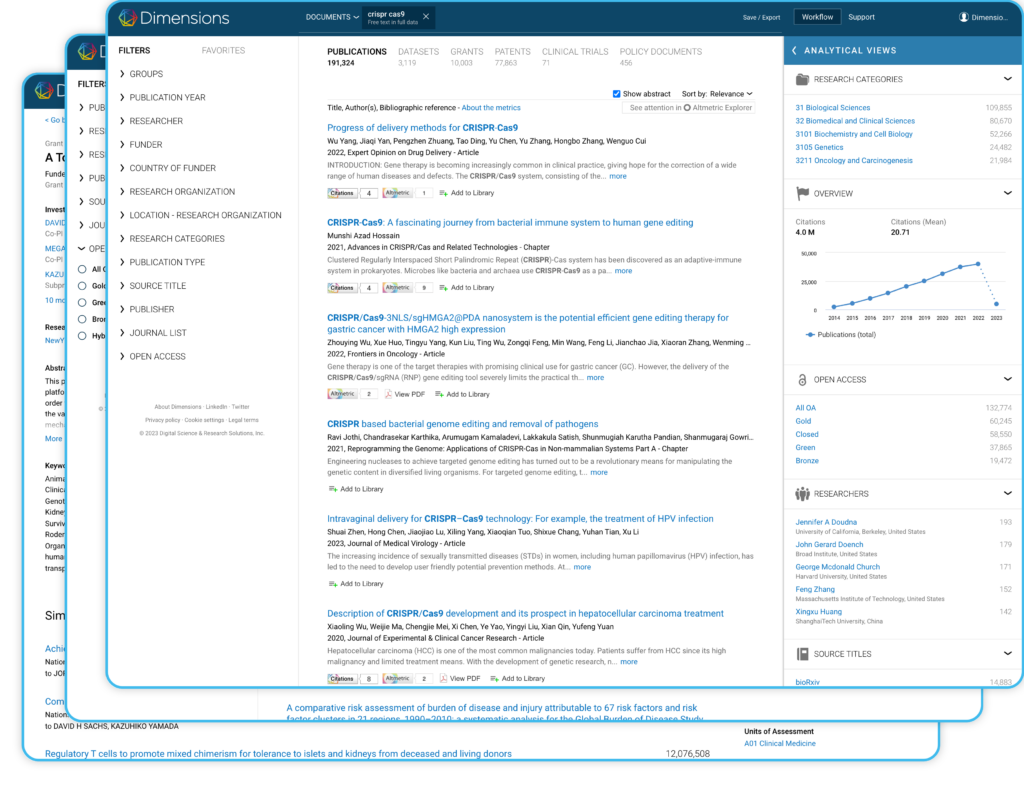
- Dimensions Analytics – our primary solution. You receive full access to features and data – one platform for research discovery and analytics
- The free version: app.dimensions.ai for non commercial use.
- Dimensions applications, which can be added to our primary solution to fast track specific tasks.
- You can also have a custom configuration according to your organization’s needs, either as a publicly available portal or with proprietary data securely integrated.



